No products in the cart.
How to calculate maximum Hydrogen output
Generation of Hydrogen is based on electrolysis, which is governed by the laws of physics.
This process was studied almost 200 years ago by Michael Faraday, who subsequently published “Faraday’s Laws of Electrolysis”. The laws state that an electrolysis cell, operating at a certain current (amps) will produce a known amount of HHO.
The two main considerations are the number of electrode plates and the actual active surface area.
The active area is the surface area of the plates minus the area of the gaskets.
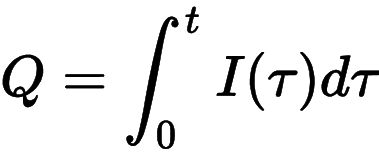
For example, if a generator has 20 cm plates and 13 mm wide gaskets:
The plate area is 20 cm X 20 cm = 400 square cm
The active area is 17.4 cm X 17.4 cm = 303 square cmThe 303 square cm number must be used for calculations.
Michael Faraday also demonstrated that electrolysis cells can support up to 0.084 amps per square cm without overheating. This is the standard used to design a HHO generator.
Therefore, the 303 square cm generator can support up to 25.4 amps of current.
The number of plates is also very important. Too few and the generator will have poor HHO production and overheat. Too many plates and the generator may not work at all.
For 12 volt vehicles, the ideal number of plates is seven, which creates six electrolysis cells within the generator.
As a mathematical simplification of Faraday’s laws, a 7 plate generator will produce 64 ml/minute of HHO per 1 amp of current
So, the generator in our example will have a maximum output of 1.6 LPM (64 ml x 25.4 amps)
Summary

Article Name
Calculation of maximum Hydrogen production, Faraday's Laws of Electrolysis
Description
Generation of Hydrogen is based on electrolysis, which is governed by the laws of physics. This process was studied almost 200 years ago by Michael Faraday, who subsequently published "Faraday's Laws of Electrolysis".
The laws state that an electrolysis cell, operating at a certain current (amps) will produce a known amount of HHO.
The two main considerations are the number of electrode plates and the actual active surface area.
Author
Better Fuel Technology
Publisher Name
Better Fuel Technology
Publisher Logo

Read more:
How hydrogen generators for vehicles work
How to select correct equipment
How to convert engines to use hydrogen as primary fuel
What happens when too much hydrogen is supplied to an engine
Setup and operation of HHO equipment, safety, compatibility
Results of NASA’ s experiments with hydrogen in internal combustion engines
Effect of Hydrogen HHO on Emissions
Visit our store
Summary

Article Name
Calculation of maximum Hydrogen production, Faraday's Laws of Electrolysis
Description
Generation of Hydrogen is based on electrolysis, which is governed by the laws of physics. This process was studied almost 200 years ago by Michael Faraday, who subsequently published "Faraday's Laws of Electrolysis".
The laws state that an electrolysis cell, operating at a certain current (amps) will produce a known amount of HHO.
The two main considerations are the number of electrode plates and the actual active surface area.
Author
Better Fuel Technology
Publisher Name
Better Fuel Technology
Publisher Logo








